24Jan, 2021
0
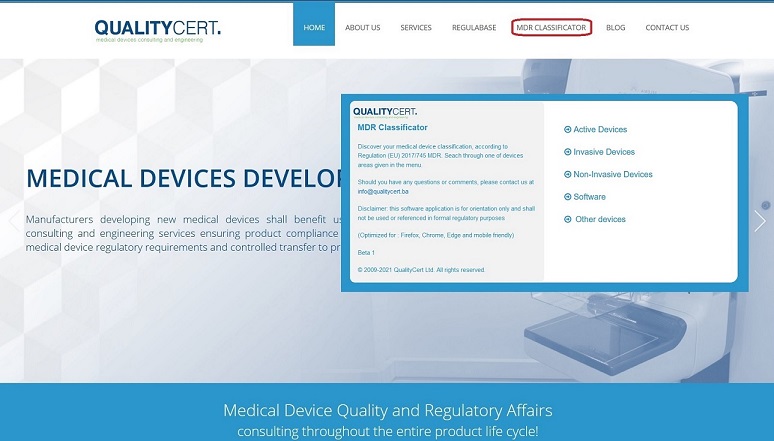
QualityCert Web App – MDR Classificator – Medical devices classification according to the EU Medical Device Regulation (EU MDR 2017/745)
MDR Classificator QualityCert Ltd. introduces unique web application, the “MDR Classificator”, provided as a tool for medical device industry stakeholders, primarily for Manufacturers, as well as others involved in regulatory processes where medical device classification is taking place. The MDR Classificator is based on the MDR (EU Medical Device Regulation 2017/745) classification requirements and rules structure. MDR Classificator has been released in Beta 1 version and it is generic questionnaire that mirrors the statements of MDR Annex VIII, Chapter III Classification rules. With a solid understanding of the classification […]
READ MORE