QualityCert Web App – MDR Classificator – Medical devices classification according to the EU Medical Device Regulation (EU MDR 2017/745)
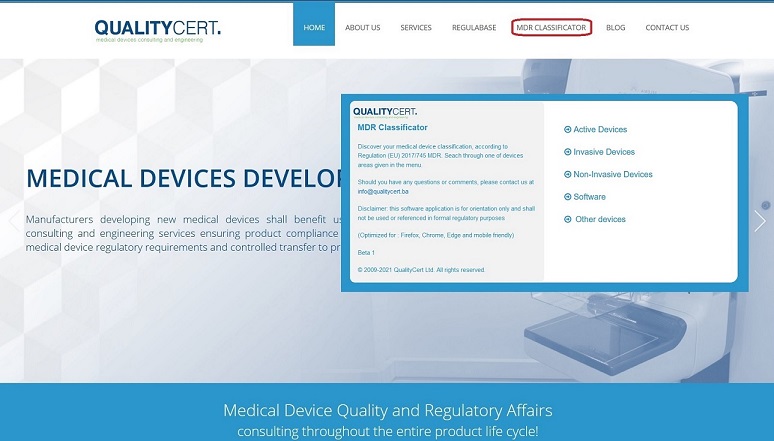
MDR Classificator
QualityCert Ltd. introduces unique web application, the “MDR Classificator”, provided as a tool for medical device industry stakeholders, primarily for Manufacturers, as well as others involved in regulatory processes where medical device classification is taking place.
The MDR Classificator is based on the MDR (EU Medical Device Regulation 2017/745) classification requirements and rules structure. MDR Classificator has been released in Beta 1 version and it is generic questionnaire that mirrors the statements of MDR Annex VIII, Chapter III Classification rules. With a solid understanding of the classification requirements and device characteristics, MDR Classificator enables user to reach the final classification result of identified medical device classification per applicable rule.
In order to better understand and facilitate the classification process of medical devices, user may utilize available MDR relevant classification guidelines.
MDR Classificator is available at www.QualityCert.ba web page Menu.
Disclaimer: QualityCert MDR Classificator is software web application for user’s orientation only and shall not be used or referenced in formal regulatory purposes.
*******************************
Classification requirements
Article 51 of EU MDR 2017/745 (MDR) requires all medical devices to be divided into four classes, taking into account the intended purpose of the devices and their inherent risks. The classification of medical devices is a “risk-based” system, depending on the vulnerability of the human body and the potential risk associated with the device.
The classification is carried out in accordance with Annex VIII of MDR and it is required in process of meeting European medical devices regulatory requirements. Once determined, the medical devices classification provides manufacturer the opportunity for selection or to follow the unique product conformity assessment (CE marking) route. Furthermore, there are numerous circumstances where such classification is required, therefore it is recommended at early stage of product design and development to determine the medical device classification.
Classification rules
According to MDR Chapter V – classification and conformity assessment, and Article 51. Classification of devices, classification levels start with Low risk (Class I) to High risk medical devices (Class III), sorted out in the following order:
Class I – Low risk medical devices
Class I(m) – Low risk medical devices with measuring function
Class I(s) – Low risk medical devices in sterile condition
Class I(r) – Low risk medical devices intended for reuse
Class IIa – Medium risk medical devices
Class IIb – Medium-high risk medical device
Class III – High risk medical devices
Medical device classification level is determined by assessment of its intended use against the applicable classification rules. Annex VIII, chapter III provides 22 classification rules broken down into the following categories:
Rules (1– 4): Non-invasive Devices
Rules (5 – 8): Invasive Devices
Rules (9 – 13): Active Devices
Rules (14 – 22): Special Rules
Comparing to departing medical devices Directive (93/42/EEC MDD), the forthcoming Medical Device Regulation (2017/745 MDR) introduces 4 new rules in the category of “Special rules” and also some MDD rules have been resorted throughout MDR classification rules structure. Additional change is made in Class I category, where new type of potential classification has been introduced for medical devices intended to be reused, which are identified as Class I(r) reusable device. Class I(m), I(s) and I(r) as such are not identified by the MDR classification, but general Class I for all. However, considering medical device intended use, condition and function, the classification status shall be appended by additional identification (m – measurement, s – sterile and r – reused), where applicable. All these three Class I categories require Notified Body conformity assessment.